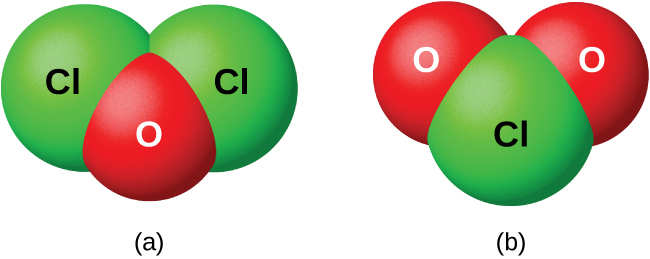

Chlorine is the second halogen, being a nonmetal in group 17 of the periodic table. Its properties are thus similar to fluorine, bromine, and iodine, and are largely. The 7 elements classified as "other metals" are located in groups 13, 14, and 15. While these elements are ductile and malleable, they are not the same as the. Bleaching sequences can be any combination of these stages. Lately selection of bleaching sequence has largely being guided by environmental considerations. Whole Lotta Rules Going On The process of naming compounds is just a set of rules. We're going to show you some of the basics. There are some advanced ways of naming. A drawing of a carbon image, and carbon fiber. Carbon fiber is extremely strong and is used as a structural material when both strength and light weight are required. Multi-Industry Uses for Oxygen: Oxygen is used with fuel gases in gas welding, gas cutting, oxygen scarfing, flame cleaning, flame hardening, and flame straightening.
Oxygen. The chemical element oxygen is classed as a chalcogen and a nonmetal. It was discovered in 1.
Joseph Priestley and and two years earlier, but unpublished, by Carl W. Scheele. Data Zone. Classification: Oxygen is a chalcogen and a nonmetal Color: colorless Atomic weight: 1. State: gas Melting point: - 2. C, 5. 4. 8 K Boiling point: - 1.

C, 9. 0. 2 K Electrons: 8. Protons: 8. Neutrons in most abundant isotope: 8.

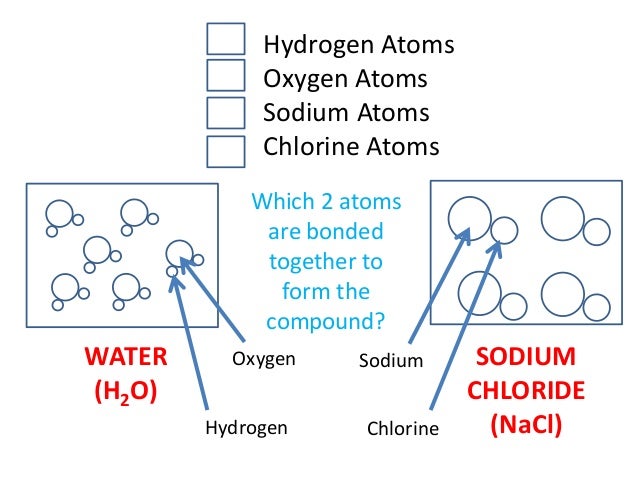
Chem4Kids.com! This tutorial introduces chemical compounds. Other sections include matter, elements, the periodic table, reactions, and biochemistry. Oxygen was discovered in 1774 by Joseph Priestley in England and two years earlier, but unpublished, by Carl W. Scheele in Sweden. Scheele heated several compounds. The millions of different chemical compounds that make up everything on Earth are composed of 118 elements that bond together in different ways. This module explores.
Electron shells: 2,6 Electron configuration: 1s. Density @ 2. 0o. C: 0. Show more, including: Heats, Energies, Oxidation, Reactions, Compounds, Radii, Conductivities. Atomic volume: 1. Structure: bcc: body- centered cubic when solid Specific heat capacity 0.
J g- 1 K- 1. Heat of fusion 0. J mol- 1 of O2. Heat of atomization 2. J mol- 1. Heat of vaporization 6. J mol- 1 of O2. 1st ionization energy 1. J mol- 1. 2nd ionization energy 3. J mol- 1. 3rd ionization energy 5.
J mol- 1. Electron affinity 1. J mol- 1. Minimum oxidation number - 2 Min. Lavoisier’s wife Marie- Anne makes notes.


She also created the engraving from which this image was taken. Oxygen cylinders. Art restoration with oxygen – comparing cleaning and restoring a painting with acetone and methylene chloride (left) vs atomic oxygen (right). Oxygen is very efficient at removing carbon based dirt. Image: NASA. Watch steel melt when charcoal (carbon) burns in liquid oxygen.
Higher concentrations lead to faster reaction rates.)Discovery of Oxygen. Oxygen was discovered in 1. Joseph Priestley in England and two years earlier, but unpublished, by Carl W. Scheele in Sweden. Scheele heated several compounds including potassium nitrate, manganese oxide, and mercury oxide and found they released a gas which enhanced combustion. Priestley’s lens was smaller than the enormous one used by Antoine Lavoisier in his investigation of carbon. Priestley wrote: “But what surprised me more than I can well express was that a candle burned in this air with a remarkably vigorous flame.
I was utterly at a loss how to account for it.” (1) In addition to noticing the effect of oxygen on combustion, Priestley later noted the new gas’s biological role. He placed a mouse in a jar of oxygen, expecting it would survive for 1. Instead, the mouse survived for a whole hour and was none the worse for it.(2) Antoine Lavoisier carried out similar experiments to Priestley’s and added to our knowledge enormously by discovering that air contains about 2.
This enabled Lavoisier to state a new fundamental law: the law of the conservation of matter; “matter is conserved in chemical reactions” or, alternatively, “the total mass of a chemical reaction’s products is identical to the total mass of the starting materials.” In addition to these achievements, it was Lavoisier who first gave the element its name oxygen. Watch it stick to a magnet. Electrical energy is used to split water. Watch out for the different ways the two gases are collected. Appearance and Characteristics. Harmful effects: O2 is non- toxic under normal conditions. However, exposure to oxygen at higher than normal pressures, e.
Ozone (O3) is toxic and if inhaled can damage the lungs. Characteristics: Oxygen in its common form (O2) is a colorless, odorless and tasteless diatomic gas. Oxygen is extremely reactive and forms oxides with nearly all other elements except noble gases. Oxygen dissolves more readily in cold water than warm water. As a result of this, our planet’s cool, polar oceans are more dense with life than the warmer, tropical oceans. Liquid and solid oxygen are pale blue and are strongly paramagnetic. Ozone (O3), another form (allotrope) of oxygen, occurs naturally in the Earth’s upper atmosphere.
It is made by the action of ultraviolet light on O2. Ozone shields us from much of the harmful ultraviolet radiation coming from the sun. In Earth’s early atmosphere, before oxygen and hence ozone levels were sufficiently high, the ultraviolet radiation reaching our planet’s surface would have been lethal to many organisms.(5) The reaction with oxygen is one of the criteria we use to distinguish between metals (these form basic oxides) and non- metals (these form acidic oxides). Uses of Oxygen The major commercial use of oxygen is in steel production. Carbon impurities are removed from steel by reaction with oxygen to form carbon dioxide gas. Plants and animals rely on oxygen for respiration.
Pure oxygen is frequently used to help breathing in patients with respiratory ailments. Abundance and Isotopes.
Abundance earth’s crust: 4. Abundance solar system: 9,0.
Cost, pure: $0. 3 per 1. Cost, bulk: $0. 0.
Source: Oxygen is the most abundant element in the Earth’s crust, accounting for almost half of it by mass. More than half of the atoms in the Earth’s crust are oxygen atoms. About 8. 6 percent of the mass of Earth’s oceans is oxygen – mainly in the form of water. Oxygen is the third most common element in the Universe, behind hydrogen and helium. It is obtained commercially from liquefied air separation plants. It can be prepared in the laboratory by electrolysis of water. Isotopes: 1. 3 whose half- lives are known, with mass numbers 1.
Naturally occurring oxygen is a mixture of its three stable isotopes and they are found in the percentages shown: 1. O (9. 9. 8%), 1. 7O (0. O (0. 2%). References 1. Francis Preston Venable: A Short History of Chemistry., (2. Bibliobazaar. 2. Leslie Alan Horvitz, Eureka!: Scientific Breakthroughs that Changed the World., (2. Leslie Alan Horvitz, Eureka!: Scientific Breakthroughs that Changed the World., (2. Mary Elvira Weeks, The discovery of the elements.
Three important gases., J. Educ., 1. 93. 2, 9 (2), p 2. John Mayow, Tractatus Quinque Medico- Physici, 1.
Online Book. 5. Malcolm Dole, The Natural History of Oxygen., The Journal of General Physiology., 1.
Glossary Term - 1. Most Abundant Elements in the Earth's Crust. Most Abundant Elements in the Earth's Crust. Source: CRC Handbook of Chemistry and Physics, 7. Edition. Element. Abundancepercent by weight. Abundanceparts per million by weight.
Oxygen. 46. 1%4. 61,0. Silicon. 28. 2%2. Aluminum. 8. 2. 3%8. Iron. 5. 6. 3%5. 6,3. Calcium. 4. 1. 5%4.
Sodium. 2. 3. 6%2. Magnesium. 2. 3. 3%2. Potassium. 2. 0. 9%2. Titanium. 0. 5. 65%5,6.
Hydrogen. 0. 1. 4%1,4.
 RSS Feed
RSS Feed
